Maintaining joint health and mobility has become a central focus in the nutraceutical industry, driven by an aging global population and rising consumer demand for preventive healthcare strategies that support an active lifestyle. According to United Nations projections, the global population aged 65 years and older is expected to reach 1.5 billion by 2050, a significant increase from 703 million in 2019, highlighting the urgent need for effective interventions to address age-related musculoskeletal decline1.
The evolution of joint and mobility actives
Historically, dietary supplementation for joint health has been dominated by well-established compounds such as glucosamine, chondroitin sulfate, and MSM (methylsulfonylmethane), which have long served as the standard of care for individuals seeking to alleviate symptoms of osteoarthritis and general joint discomfort2,3.
Traditional approaches to joint health
The foundational paradigm of joint supplementation has relied heavily on structural molecules intended to provide cartilage with the substrates needed for repair and maintenance, primarily through long-term administration of glycosaminoglycans and amino sugars. While these symptomatic, slow-acting drugs have shown potential for pain reduction and functional improvement in osteoarthritis patients, growing interest in nutraceutical supplements has broadened the scope to include a heterogeneous class of molecules that modulate inflammation, oxidative stress, and cartilage synthesis beyond simple substrate provision2,4.
Emerging trends in supplement formulation
Contemporary formulation strategies are increasingly shifting toward multi-targeted approaches that integrate bioactive peptides, advanced botanical extracts, and microbiome-modulating ingredients to address the complex pathophysiology of osteoarthritis through anti-inflammatory, antioxidant, and tissue-protective mechanisms2,5.
Next-generation actives: mechanisms and evidence
This section examines the scientific basis for innovative bioactive compounds that target specific pathophysiological pathways involved in joint degeneration, moving beyond substrate provision to modulate inflammatory mediators and cartilage metabolism. Among these advancements, hydrolyzed collagen and low-molecular-weight collagen peptides have garnered significant attention for their ability to deliver bioactive sequences that reach joint tissues and exert chondroprotective effects6,7.
Novel peptides and proteins
Collagen hydrolysates (Figure 1), characterized by low-molecular-weight peptides typically 3 – 6 kDa, represent a significant advancement in protein-based ingredients because of their potential to accumulate in joint tissue and stimulate cartilage repair8,9. Clinical trials of these collagen derivatives have shown evidence of efficacy in reducing pain and improving joint function, although their ability to treat and reverse advanced joint degeneration remains to be fully established2. Beyond collagen derivatives, emerging research is investigating the role of bioactive peptides derived from other protein sources, such as casein and whey, which may exert anti-inflammatory effects by modulating pro-inflammatory cytokines and oxidative stress pathways8,10. These bioactive sequences have been shown to accumulate in articular cartilage and to stimulate extracellular matrix synthesis, providing a mechanistic rationale for their use in osteoarthritis management11,12,13.
Figure 1 Processing of native collagen into small low-molecular-weight peptides
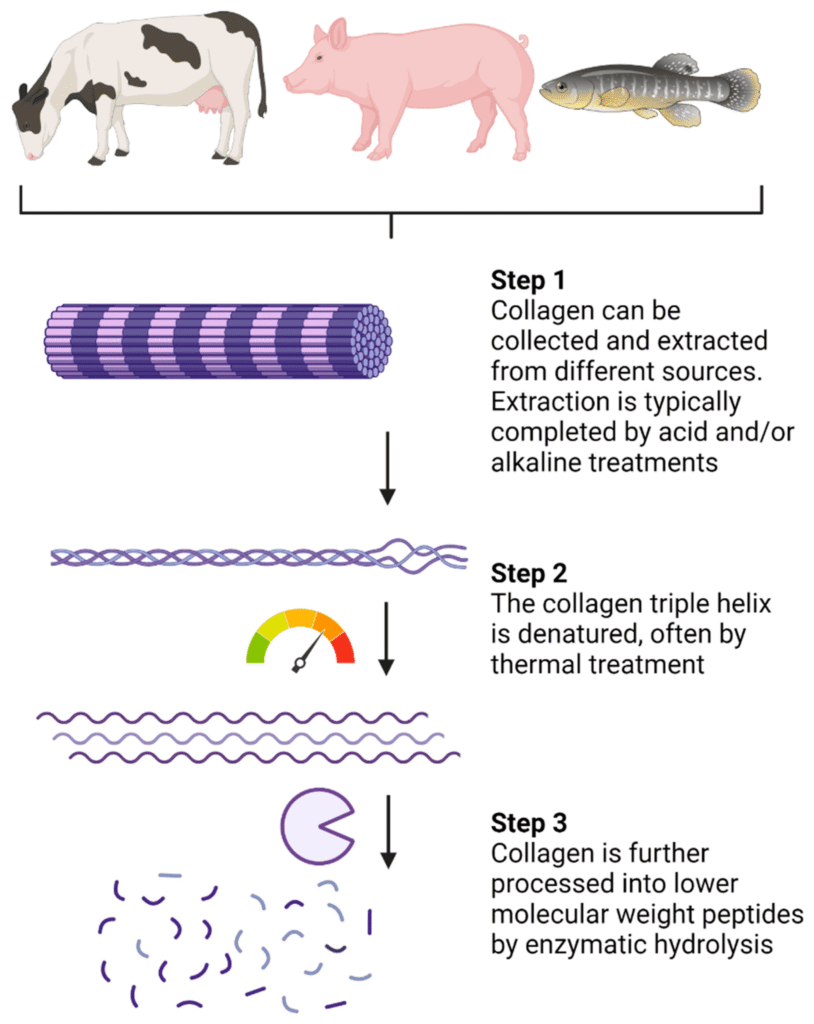
Created with Biorender.com.
Taken from: Larder, Christina E., et al. “Collagen Hydrolysates: A Source of Bioactive Peptides Derived from Food Sources for the Treatment of Osteoarthritis.” Medicines, vol. 10, no. 9, 2023, p. 50, https://doi.org/10.3390/medicines10090050
Advanced botanical extracts
Parallel to the development of novel protein-based ingredients, advanced botanical extracts are increasingly utilized for their potent anti-inflammatory and antioxidant properties, offering a complementary strategy to traditional substrate provision in osteoarthritis management16. Compounds such as hydroxytyrosol, a phenolic component found in olive leaf and oil, have demonstrated the capacity to inhibit prostaglandin E2 and nitric oxide production pathways, thereby attenuating the inflammatory cascade associated with joint generation15. Similarly, curcuminoids derived from turmeric and polyphenols from other plant sources have been investigated for their capacity to downregulate nuclear factor-kappa B signaling and reduce matrix metalloproteinase expression, thereby protecting cartilage from inflammatory degradation16. Studies indicate that supplementation with these polyphenol-rich botanical extracts can decrease serum levels of tumor necrosis factor-alpha and matrix metalloproteinase-3 in synovial fluid, while cellular models suggest they inhibit inflammatory pathways by reducing the production of inducible nitric oxide synthase and cyclooxygenase-2 enzymes17. This multi-pathway inhibition is further exemplified by ginger root extract, whose active constituent 6-gingerol exerts anti-inflammatory effects by reducing inducible nitric oxide synthase and TNF-α expression through the blocking of NF-κB and protein kinase C signaling in LPS-stimulated macrophages18. Boswellia serrata extracts, specifically those containing 5-Loxin and Aflapin®, further contribute to this anti-inflammatory landscape by reducing the enzymatic degradation of cartilage and modulating pro-inflammatory mediators19. Recent systematic reviews have highlighted that these phytochemicals, including curcumin and boswellic acid, may offer superior efficacy compared to traditional nutraceuticals such as glucosamine and chondroitin alone in managing osteoarthritis symptoms20,21.
Microbiome-targeted interventions
The gut-joint axis represents a promising frontier in next-generation supplementation, predicated on the understanding that intestinal dysbiosis and systemic inflammation contribute significantly to the osteoarthritis pathogenesis. Preclinical evidence suggests that modulating the gut microbiome through probiotic and prebiotic interventions can reduce systemic levels of inflammatory cytokines such as interleukin-1β and tumor necrosis factor-alpha, thereby potentially attenuating joint destruction22,23. Clinical studies support this mechanistic link, demonstrating that probiotic strains such as Lactobacillus casei Shirota can significantly lower C-reactive protein levels while improving pain and functional scores in patients with knee osteoarthritis23(Figure 2).
Figure 2 Mechanism of bioactives actions in modulating the bone metabolism of osteoarthritis by gut-immune-bone axis
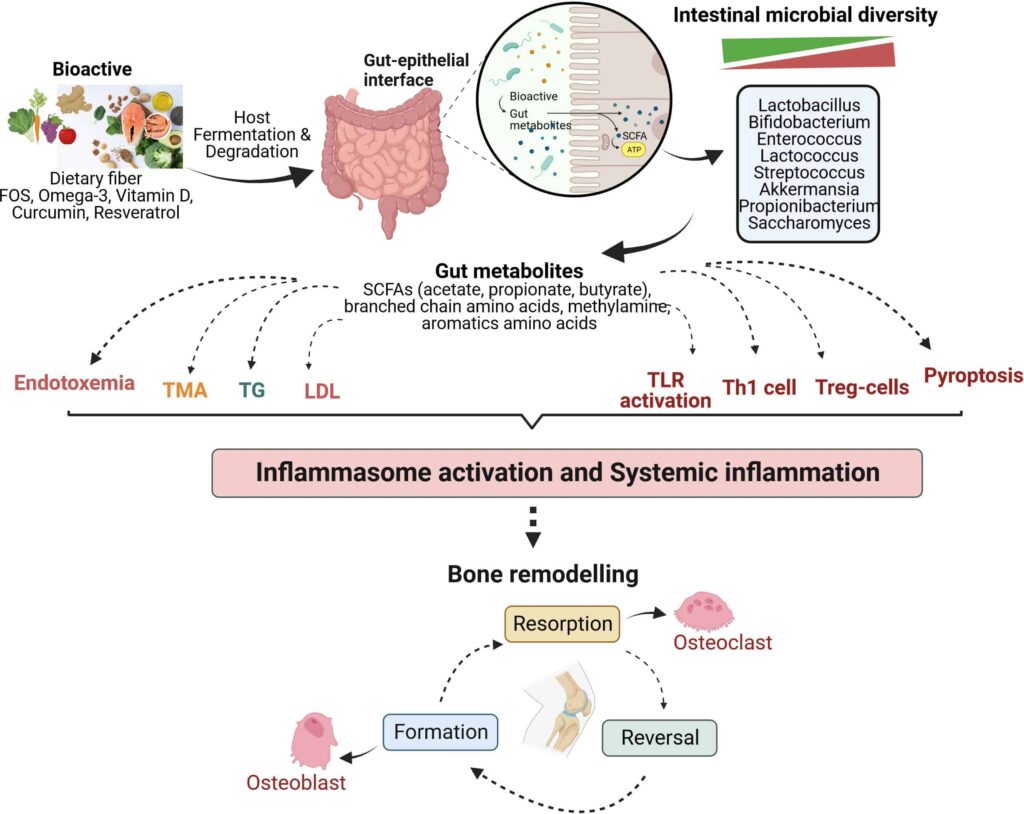
Various bioactive compounds undergo fermentation in the host gut by intestinal microbiota, thereby regulating microbial diversity and generating metabolites that majorly modulate the immune system, among other significant functions. They are involved in lipid metabolism, influencing the levels of triglycerides and low-density lipoprotein. Gut metabolites also lead to differentiation and activation of immune cells to strengthen the intestinal barrier and maintain gut homeostasis. These mechanisms act at the system level, reducing inflammation and affecting bone remodelling to balance osteoclastogenesis (bone resorption) and osteoblastogenesis (bone formation). FOS, Fructooligosaccharide; SCFAs, Short chain fatty acids; TMA, Trimethylamine; TG, Triglyceride; LDL, Low density lipoprotein; TLR, Toll-like receptor; Th1, Helper T lymphocytes type 1; Treg, Regulatory T lymphocytes.
Taken from Basak, Sanjay, et al. “Bioactives and Their Roles in Bone Metabolism of Osteoarthritis: Evidence and Mechanisms on Gut-bone Axis.” Frontiers in Immunology, vol. 14, 2024, p. 1323233, https://doi.org/10.3389/fimmu.2023.1323233
Building upon these individual probiotic effects, research indicates that combining probiotic complexes with bioactive compounds such as rosavin and zinc can upregulate tissue inhibitors of metalloproteinases while downregulating matrix metalloproteinases in human chondrocytes, thereby offering a multi-faceted approach to cartilage protection. Emerging evidence suggests that established nutraceuticals such as glucosamine sulfate and green-lipped mussel extract may also exert therapeutic benefits by regulating intestinal microbiota composition and immunity, with specific reductions in Clostridia species correlating with symptomatic improvement24.
Synergistic ingredient combinations
The strategic combination of bioactive compounds has emerged as a critical approach to enhance therapeutic efficacy, as preclinical models and clinical trials suggest that mixtures such as curcumin and boswellic acids exhibit additive synergistic activity in the management of osteoarthritis pain25,26. This multi-targeted approach is further supported by evidence indicating that formulations combining undenatured type II collagen with hyaluronic acid and cat’s claw extract can decrease pain and improve physical function with negligible adverse events, particularly when integrated with conventional symptomatic treatments and physical therapy17. Furthermore, recent preclinical data indicate that co-administration of specific probiotic strains with traditional substrates such as type II collagen and glucosamine can significantly inhibit pro-inflammatory cytokines, including TNF-α, IL-1β, and IL-6, while upregulating anti-inflammatory mediators such as IL-10 and TGF-β, resulting in reduced cartilage destruction compared to single-agent interventions24. This synergistic efficacy is further corroborated by clinical evidence demonstrating that combining glucosamine with omega-3 fatty acids, chondroitin, or curcumin yields superior pain reduction and symptom management in knee osteoarthritis compared to monotherapies27.
Formulation challenges and opportunities
Despite the promising therapeutic potential of these next-generation bioactive compounds, their successful translation into effective food supplements depends on overcoming significant formulation hurdles related to physicochemical stability and physiological delivery.
Bioavailability and absorption enhancement
The therapeutic efficacy of many next-generation actives, particularly polyphenols such as curcumin and lipophilic compounds, is often limited by their poor aqueous solubility and extensive first-pass metabolism, necessitating advanced delivery systems to achieve adequate tissue concentrations28. Novel delivery technologies, such as phytosome complexes, micelles, and nanoparticle emulsions, have been developed to overcome these limitations by enhancing solubility and protecting active ingredients from premature degradation in the gastrointestinal tract29,30. For instance, the incorporation of hydrolyzed collagen type II with low-molecular-weight hyaluronic acid and chondroitin sulfate creates a naturally occurring matrix that mimics human articular cartilage composition, thereby facilitating improved absorption and retention in synovial joints31. Formulation science must also address the pharmacokinetic parameters of absorption, distribution, metabolism, and excretion, as the manufacturing process and delivery vehicle can significantly alter the bioavailability and therapeutic efficacy of nutraceuticals32.

Consumer acceptance and palatability
Beyond physiological efficacy, the commercial success of joint and mobility supplements is fundamentally dependent on organoleptic properties and patient compliance, as many bioactive ingredients, such as marine-derived compounds and concentrated botanical extracts, possess inherent bitter tastes, unpleasant odors, or gritty textures that deter consistent consumption. To mitigate these sensory defects, manufacturers are increasingly employing advanced encapsulation technologies, such as lipid-based microencapsulation and spray-drying techniques, which effectively mask unpleasant flavors and odors while protecting the bioactive core from environmental degradation33.
Market trends
The commercial viability of next-generation joint and mobility supplements is heavily influenced by the complex interplay between evolving regulatory standards and dynamic market forces across global jurisdictions. These frameworks vary significantly in their classification of bioactive ingredients, with some jurisdictions permitting structure-function claims based on traditional use while others require rigorous pharmaceutical-grade clinical trials to substantiate efficacy and safety34. Bioactive compounds are inherently susceptible to degradation from environmental factors such as heat, pH fluctuations, oxidation, light exposure, and hydrolysis, necessitating careful optimization of processing and storage methods to preserve molecular integrity in the final product35. Oxidation and hydrolytic degradation pose significant risks to the potency of sensitive ingredients like polyphenols and unsaturated fatty acids, requiring the use of stabilizing agents and protective packaging to maintain shelf-life36,37. Nanotechnology-driven approaches, including liposomes, microparticles, and nanoparticles, offer promising solutions by providing controlled release and enhanced protection against the harmful biological milieu that typically compromises bioavailability38. In vitro studies have demonstrated that, in osteoarthritis, the phospholipid layer, acting as a boundary lubricant, was absent from the articular surface of degenerated cartilage, highlighting the need for formulations that can restore this protective function38.
Consumer demand and market growth projections
The global market for nutraceuticals and dietary supplements has experienced a remarkable surge in demand over the past decade, driven by a growing emphasis on preventive healthcare and heightened consumer preference for bioactive products. Market analyses project substantial growth in the joint health sector, with the collagen market alone estimated to reach $6.63 billion by 2025 and U.S. consumers expected to spend approximately $122 million on collagen products, a significant portion of which will be allocated to collagen-based nutraceuticals for bones and joints32. Ethical considerations in product development require rigorous quality assurance and transparency to address consumer skepticism regarding safety, efficacy, and the accurate labeling of bioactive ingredients. The absence of unified international standards for nanomaterials and novel delivery systems creates significant barriers to commercialization, as regulatory bodies such as the European Food Safety Authority and the U.S. Food and Drug Administration increasingly mandate comprehensive toxicological testing and transparent labelling practices to ensure consumer safety39.
Conclusion
The advancement of joint and mobility actives in food supplements marks a paradigm shift from traditional symptomatic relief toward multifunctional, evidence-based interventions that target the underlying pathophysiology of musculoskeletal degeneration. By integrating novel bioactive compounds, such as advanced peptides, targeted botanical extracts, and microbiome-modulating agents, with sophisticated delivery technologies, next-generation formulations have the potential not only to alleviate pain but also to modify disease progression and restore functional capacity38,40. However, the successful translation of these scientific advancements into mainstream clinical practice depends on addressing persistent methodological limitations in current research and establishing robust, standardized clinical evidence that can withstand regulatory scrutiny32,41. Future research directions must prioritize large-scale, randomized controlled trials that use validated biomarkers and standardized outcome measures to definitively establish the therapeutic efficacy and safety profiles of these emerging ingredients32,42.
References:
- Yves, H., Herman, J., Uebelhoer, M. et al. Oral supplementation with fish cartilage hydrolysate in an adult population suffering from knee pain and function discomfort: results from an innovative approach combining an exploratory clinical study and an ex vivo clinical investigation. BMC Musculoskelet Disord 24, 748 (2023). https://doi.org/10.1186/s12891-023-06800-4
- Colletti, A.; Cicero, A.F.G. Nutraceutical Approach to Chronic Osteoarthritis: From Molecular Research to Clinical Evidence. Int. J. Mol. Sci. 2021, 22, 12920. https://doi.org/10.3390/ijms222312920
- Mobasheri, A., Mahmoudian, A., Kalvaityte, U. et al. A White Paper on Collagen Hydrolyzates and Ultrahydrolyzates: Potential Supplements to Support Joint Health in Osteoarthritis?. Curr Rheumatol Rep 23, 78 (2021). https://doi.org/10.1007/s11926-021-01042-6
- Cimini, Donatella, et al. “Evaluation of Unsulfated Biotechnological Chondroitin in a Knee Osteoarthritis Mouse Model As a Potential Novel Functional Ingredient in Nutraceuticals and Pharmaceuticals.” Frontiers in Bioengineering and Biotechnology, vol. 10, 2022, p. 934997, https://doi.org/10.3389/fbioe.2022.934997
- Maouche, Ahmed, et al. “Bioactive Compounds in Osteoarthritis: Molecular Mechanisms and Therapeutic Roles.” International Journal of Molecular Sciences, vol. 25, no. 21, 2023, p. 11656, https://doi.org/10.3390/ijms252111656
- Park, Sun, et al. “Efficacy and Safety of Low-molecular-weight Collagen Peptides in Knee Osteoarthritis: A Randomized, Double-blind, Placebo-controlled Trial.” Frontiers in Nutrition, vol. 12, 2025, p. 1644899, https://doi.org/10.3389/fnut.2025.1644899.
- “Collagen Supplementation for Joint Health: The Link between Composition and Scientific Knowledge.” Nutrients, vol. 15, no. 6, 2022, p. 1332, https://doi.org/10.3390/nu15061332.
- Honvo, G., Lengelé, L., Charles, A. et al. Role of Collagen Derivatives in Osteoarthritis and Cartilage Repair: A Systematic Scoping Review With Evidence Mapping. Rheumatol Ther 7, 703–740 (2020). https://doi.org/10.1007/s40744-020-00240-5
- Larder, Christina E., et al. “Collagen Hydrolysates: A Source of Bioactive Peptides Derived from Food Sources for the Treatment of Osteoarthritis.” Medicines, vol. 10, no. 9, 2023, p. 50, https://doi.org/10.3390/medicines10090050
- Mobasheri, A., Mahmoudian, A., Kalvaityte, U. et al. A White Paper on Collagen Hydrolyzates and Ultrahydrolyzates: Potential Supplements to Support Joint Health in Osteoarthritis?. Curr Rheumatol Rep 23, 78 (2021). https://doi.org/10.1007/s11926-021-01042-6
- Dobenecker, Britta, et al. “The Oral Intake of Specific Bioactive Collagen Peptides (BCP) Improves Gait and Quality of Life in Canine Osteoarthritis Patients—A Translational Large Animal Model for a Nutritional Therapy Option.” PLOS ONE, vol. 19, no. 9, 2024, p. e0308378, https://doi.org/10.1371/journal.pone.0308378.
- Lin, CR., Tsai, S.H.L., Huang, KY. et al. Analgesic efficacy of collagen peptide in knee osteoarthritis: a meta-analysis of randomized controlled trials. J Orthop Surg Res 18, 694 (2023). https://doi.org/10.1186/s13018-023-04182-w.
- Yuenyongviwat, Varah, et al. “Efficacy of Combined Undenatured Type II Collagen and Hydrolysed Collagen Supplementation in Knee Osteoarthritis: A Randomised Controlled Trial.” Scientific Reports, vol. 15, no. 1, 2025, p. 32313, https://doi.org/10.1038/s41598-025-17505-0.
- Liu, Xiaoqian, et al. “Which Supplements Can I Recommend to My Osteoarthritis Patients?” Rheumatology, vol. 57, no. suppl_4, 2018, pp. iv75-iv87, https://doi.org/10.1093/rheumatology/key005.
- Mével, Elsa, et al. “Olive and Grape Seed Extract Prevents Post-traumatic Osteoarthritis Damages and Exhibits in Vitro anti IL-1β Activities Before and After Oral Consumption.” Scientific Reports, vol. 6, no. 1, 2016, p. 33527, https://doi.org/10.1038/srep33527.
- Park, Sun, et al. “Efficacy and Safety of Low-molecular-weight Collagen Peptides in Knee Osteoarthritis: A Randomized, Double-blind, Placebo-controlled Trial.” Frontiers in Nutrition, vol. 12, 2025, p. 1644899, https://doi.org/10.3389/fnut.2025.1644899.
- Aghamohammadi, Dawood, et al. “Nutraceutical Supplements in Management of Pain and Disability in Osteoarthritis: A Systematic Review and Meta-analysis of Randomized Clinical Trials.” Scientific Reports, vol. 10, no. 1, 2020, p. 20892, https://doi.org/10.1038/s41598-020-78075-x. Accessed 10 Feb. 2026.https://doi.org/10.1038/s41598-020-78075-x
- Yamagishi, Y., Someya, A., Imai, K., Nagao, J., Nagaoka, I.”Evaluation of the anti-inflammatory actions of various functional food materials including glucosamine on synovial cells”. Molecular Medicine Reports 16.
- Ganguly, A., & Banerjee, S. K. (2023). Exploring the biomarker of collagen degradation in cartilage for osteoarthritis management: The power of topical phytotherapy. International Journal of Health Sciences and Research, 13(4), 186–203 https://doi.org/10.52403/ijhsr.20230423
- Comblain, Fanny, et al. “Curcuminoids Extract, Hydrolyzed Collagen and Green Tea Extract Synergically Inhibit Inflammatory and Catabolic Mediator’S Synthesis by Normal Bovine and Osteoarthritic Human Chondrocytes in Monolayer.” PLOS ONE, vol. 10, no. 3, 2015, p. e0121654, https://doi.org/10.1371/journal.pone.0121654.
- Lewis, R., Gómez Álvarez, C.B., Rayman, M. et al. Strategies for optimising musculoskeletal health in the 21st century. BMC Musculoskelet Disord 20, 164 (2019). https://doi.org/10.1186/s12891-019-2510-7
- Mével, Elsa, et al. “Olive and Grape Seed Extract Prevents Post-traumatic Osteoarthritis Damages and Exhibits in Vitro anti IL-1β Activities Before and After Oral Consumption.” Scientific Reports, vol. 6, no. 1, 2016, p. 33527, https://doi.org/10.1038/srep33527. https://doi.org/10.1038/srep33527
- Basak, Sanjay, et al. “Bioactives and Their Roles in Bone Metabolism of Osteoarthritis: Evidence and Mechanisms on Gut-bone Axis.” Frontiers in Immunology, vol. 14, 2024, p. 1323233, https://doi.org/10.3389/fimmu.2023.1323233
- Sun, Chang, et al. “The Immune Role of the Intestinal Microbiome in Knee Osteoarthritis: A Review of the Possible Mechanisms and Therapies.” Frontiers in Immunology, vol. 14, 2023, p. 1168818, https://doi.org/10.3389/fimmu.2023.1168818. 2026.
- Staff, U. (2022) Nutraceutical Treatment in Knee Osteoarthritis Arthritis-Presentation of a Dietary Plan by Means of Additional Nutraceuticals in Lieu of the Detrimental Side Effects of Current Conventional Medicine. Open Journal of Orthopedics, 12, 303-326. doi: 10.4236/ojo.2022.127030.
- Sethi, Vidhu, et al. “Potential Complementary and/or Synergistic Effects of Curcumin and Boswellic Acids for Management of Osteoarthritis.” Therapeutic Advances in Musculoskeletal Disease, 2022, https://doi.org/10.1177/1759720X221124545. Accessed 11 Feb. 2026.https://doi.org/10.1177/1759720×221124545
- Sumsuzzman, Dewan M., et al. “Comparative Efficacy of Glucosamine-Based Combination Therapies in Alleviating Knee Osteoarthritis Pain: A Systematic Review and Network Meta-Analysis.” Journal of Clinical Medicine, vol. 13, no. 23, 2023, p. 7444, https://doi.org/10.3390/jcm13237444.
- Stevanny, Bella, Tungki Pratama Umar, Junoretta Haviva Ernanto, and Rachmat Hidayat. “Peanut (Arachis hypogaea) Shells Extract and Apis dorsata Honey Reduce Matrix Metalloproteinase-3 in Monosodium Iodoacetate-Induced Osteoarthritic Rats.” Journal of Biomedicine and Translational Research, vol. 6, no. 3, 31 Dec. 2020, pp. 74-78 ,https://doi.org/10.14710/jbtr.v6i3.7908
- Maouche, Ahmed, et al. “Bioactive Compounds in Osteoarthritis: Molecular Mechanisms and Therapeutic Roles.” International Journal of Molecular Sciences, vol. 25, no. 21, 2023, p. 11656, https://doi.org/10.3390/ijms252111656.
- Yabas, Mehmet, et al. “A Next Generation Formulation of Curcumin Ameliorates Experimentally Induced Osteoarthritis in Rats via Regulation of Inflammatory Mediators.” Frontiers in Immunology, vol. 12, 2021, p. 609629, https://doi.org/10.3389/fimmu.2021.609629.
- Effect of an oral preparation containing hyaluronic acid, chondroitin sulfate, hydrolyzed collagen type II and hydrolyzed keratin on synovial fluid features and clinical indices in knee osteoarthritis. A pilot study. Reumatismo [Internet]. 2020 Nov. 19 [cited 2026 Feb. 11];72(3):125-30. https://www.reumatismo.org/reuma/article/view/1272
- Mobasheri, A., Mahmoudian, A., Kalvaityte, U. et al. A White Paper on Collagen Hydrolyzates and Ultrahydrolyzates: Potential Supplements to Support Joint Health in Osteoarthritis?. Curr Rheumatol Rep 23, 78 (2021). https://doi.org/10.1007/s11926-021-01042-6
- Zhang, P., Li, K., Kamali, A. et al. Small molecules of herbal origin for osteoarthritis treatment: in vitro and in vivo evidence. Arthritis Res Ther 24, 105 (2022).https://doi.org/10.1186/s13075-022-02785-y
- Sunil Katoch, Raju Easwaran, Hemant Thacker, Amulya Kumar Singh, Pradeep Moonot, Amitava N Mukherjee, Santosh Shetty, Kaushal C Malhan, Vasudev Prabhu, Niranjan R Rathod, Moazzam Jah, Sridhar Gangavarapu, Naidu MJ, Pranay Bhushan and Anish Desai. Expert consensus on the role of nutraceuticals in bone, joints, and muscle health. Int. J. Orthop. Sci. 2022;8(3):286-292. DOI: 10.22271/ortho.2022.v8.i3e.3211
- de Castro Cogle K, Kubo MTK, Merlier F, Josse A, Anastasiadi M, Mohareb FR, Rossi C. Probabilistic Modelling of the Food Matrix Effects on Curcuminoid’s In Vitro Oral Bioaccessibility. Foods. 2024; 13(14):2234. https://doi.org/10.3390/foods1314223
- Ola, Monika, et al. “Formulation and Delivery Issues for Active Ingredients of Herbal Medicines, Nutraceuticals, and Cosmetics: Comparisons to Small-Molecule Drugs.” pp. 163-191, https://doi.org/10.1002/9781119769484.ch6.
- Kyriakoudi, Anastasia, et al. “Innovative Delivery Systems Loaded with Plant Bioactive Ingredients: Formulation Approaches and Applications.” Plants, vol. 10, no. 6, 2021, p. 1238, https://doi.org/10.3390/plants10061238.
- Craciunescu, O.; Icriverzi, M.; Florian, P.E.; Roseanu, A.; Trif, M. Mechanisms and Pharmaceutical Action of Lipid Nanoformulation of Natural Bioactive Compounds as Efficient Delivery Systems in the Therapy of Osteoarthritis. Pharmaceutics 2021, 13, 1108. https://doi.org/10.3390/pharmaceutics13081108
- Chellamboli, C., Muthamizhi, K., Ramalakshmi, S. et al. Recent advances in agri-food waste valorisation and nanoencapsulation of bioactive compounds for sustainable development. Discov Food 5, 355 (2025). https://doi.org/10.1007/s44187-025-00613-1
- Bordon, Gregor, et al. “Harnessing the Multifunctionality of Lipid-based Drug Delivery Systems for the Local Treatment of Osteoarthritis.” Biomedicine & Pharmacotherapy, vol. 168, 2023, p. 115819, https://doi.org/10.1016/j.biopha.2023.115819.
- Davidson, Rose K., et al. “Identifying Chondroprotective Diet-derived Bioactives and Investigating Their Synergism.” Scientific Reports, vol. 8, no. 1, 2018, p. 17173, https://doi.org/10.1038/s41598-018-35455-8.
- Bischof, K., Moitzi, A.M., Stafilidis, S. et al. Impact of Collagen Peptide Supplementation in Combination with Long-Term Physical Training on Strength, Musculotendinous Remodeling, Functional Recovery, and Body Composition in Healthy Adults: A Systematic Review with Meta-analysis. Sports Med 54, 2865–2888 (2024). https://doi.org/10.1007/s40279-024-02079-0




