Consumers are increasingly turning to the world of botanicals and botanical extracts as a means of safeguarding their health and wellbeing in a natural way. Among their many health benefits, these wholesome ingredients can support digestion, cardiovascular function, cognitive health, skin condition and immunity. Peppermint oil, for instance, is widely used for digestive complaints [1]; valerian and hops are known to help aid restful sleep [2] [3], and blueberry oil can deliver antioxidant benefits for the brain, heart, eyes, skin and more [4].
Consumers love botanicals because they are natural, and they fit with the wellness trend towards conscious living. However, many plant-derived ingredients cannot simply be incorporated into foods because of their poor solubility, stability, or activity characteristics [5]. Instead, one of the easiest, safest and most popular ways of harnessing their benefits is in dietary supplement form.
Globally, according to Innova Market Insights, between 2020 and 2024, the number of supplement launches containing botanicals outpaced similar launches in the food and beverage markets. The number of new botanical-containing supplements grew by 6 percent, while food and beverage launches were considerably fewer, at 4 percent and 3 percent respectively [6].
Supplements are the ideal way to preserve the efficacy and functionality of botanical ingredients, and softgel capsules are the ideal delivery format. For consumers, softgels are visually appealing and often easier to swallow than tablets. For manufacturers, they help to protect nutritionally valuable fill ingredients from oxygen, light and moisture. Data from Fact.MR shows that softgels account for more than half of the dietary supplements market. Significant expansion is predicted too, with a 7 per cent CAGR growth forecast between 2022 and 2032 [7]. Most softgels currently on the market (around 75 percent) are made from gelatin [8].
Gelatin is a versatile natural ingredient that has been used for centuries in a host of food, nutrition and healthcare applications. However, when it comes to botanical ingredients, these are often highly reactive, and they can present challenges when encapsulated in gelatin soft capsules. The most common of these is crosslinking, a natural phenomenon that causes issues with stability and dissolution, and can lead to loss of product quality, stability and efficacy.
What is crosslinking?
Crosslinking is a significant hurdle for softgel manufacturers. It is a physical or chemical process in which previously unlinked gelatin molecules become linked and bound together. These bonds generally change the macroscopic properties of the gelatin and therefore of the softgel capsule. It is usually impossible to reverse crosslinking reactions.
There are two different types of crosslinking:
• Self-crosslinking: the gelatin in the softgel shell reacts with itself as the lysine side chains of its amino acids bond with its carboxylic acid. This typically occurs during prolonged periods of storage, and can be exacerbated by external factors such as heat and humidity.
• Intra-crosslinking: active ingredients in the fill react with the amino acids in the gelatin. Some fills are more likely to cause problems than others.
When crosslinking occurs, it can dramatically increase the size of the protein chains in the gelatin shell of a softgel. A water-insoluble membrane known as a pellicle may form, impeding the release of the fill and hampering the way the softgel dissolves after consumption. This, in turn, reduces the bioavailability and effectiveness of the active ingredients.
Unfortunately, many botanicals contain substances that are extremely likely to trigger crosslinking reactions.
For example, polyphenols like flavonoids and tannins are potential crosslinkers. These are found in numerous botanical ingredients, including blueberries, cranberries, elderberries, citrus fruits, grapes, tea, cocoa, flaxseed, sesame seeds and sunflower seeds. Aldehydes, ketones and esters also exhibit crosslinking activity when encapsulated in softgels. These are found in essential oils like peppermint, lavender and eucalyptus as well as cinnamon, thyme, oregano, grapes and berries.
Based on many years of experience with different levels of crosslinking activity exhibited by various types of fill ingredients, the experts in the GELITA Pharma Institute have classified the likelihood of reactions in Table 1, below.
Table 1: Crosslinking index: Fill components with known crosslinking activity
| Functional groups | Triphenols | Polyphenols | Diphenols | Aldehydes | Ketones | Acetals, Enamines,Aminals | Esters(Amine scavengers) |
| Crosslinking Index | 10 | 9-10 | 8 | 6 | 4 | 3 | 1-2 |
How to prevent crosslinking
With many decades of experience in gelatin science, GELITA has developed a portfolio of products that enable manufacturers to overcome crosslinking, even when faced with most potentially reactive fills.
“The RXL® portfolio allows formulators to explore and harness the many exciting opportunities in nutritional supplementation based on botanical ingredients,” says Dr. Katrin Kaspar, Technical Customer Management Pharma EMEIA at GELITA. “And the latest addition to the range, RXL® Ultra, is now setting a new gold standard.”
RXL® gelatins effectively protect themselves – and softgel capsules – from crosslinking. The portfolio comprises four patent-protected products suitable for different levels of fill reactivity. All offer superior anti-crosslinking performance compared to standard gelatin, and all offer peace of mind that crosslinking will not affect product stability, efficacy or performance. RXL® gelatins are also easy to process on standard softgel equipment.
The first three products in the portfolio, RXL® Prime, RXL® Advanced and RXL® Pro, are made from specially tailored gelatin that contains small amounts of non-gelling super-hydrolysate gelatin. They offer ascending levels of protection against crosslinking, making them suitable for varying levels of fill reactivity. Being composed of pure gelatin, they are suitable for use in food supplements, as well as prescription and OTC healthcare products, and are already being used in many successful commercial products.
RXL® Ultra: the new gold standard
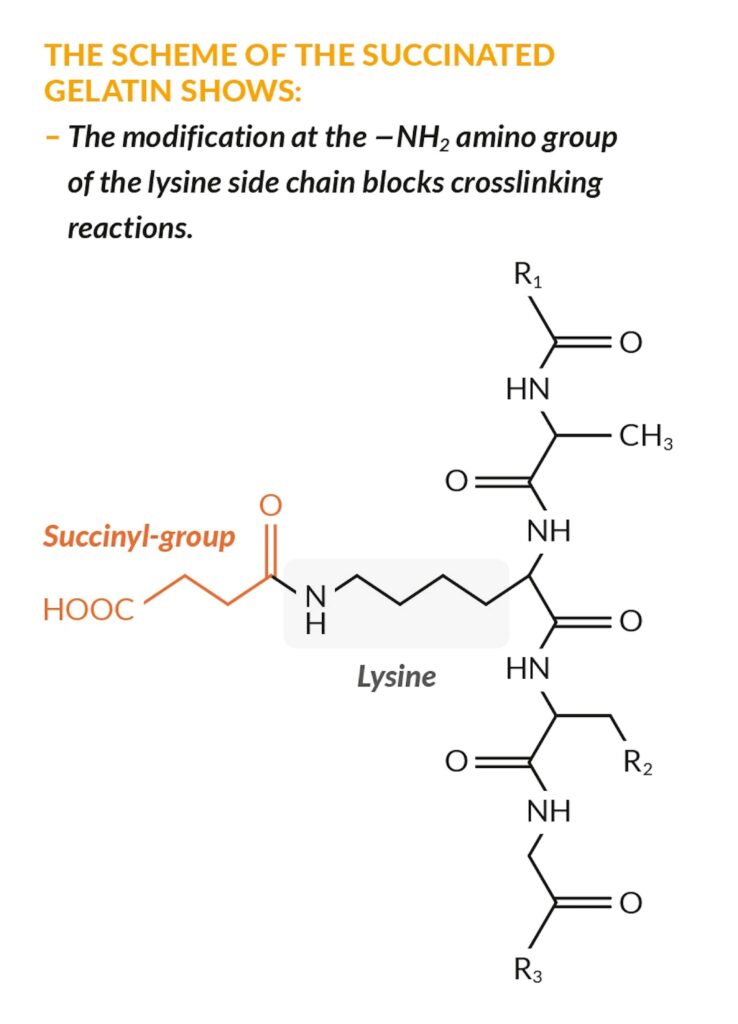
The fourth and newest addition to the range is RXL® Ultra, which is made from gelatin polysuccinate, a modified gelatin with maximised anti-crosslinking properties.
RXL® Ultra can mask the amino acid functions in the gelatin with succinic acid so that up to zero self-crosslinking or intra-crosslinking can occur. It offers excellent fill release and long-term stability for extreme crosslinking ingredients while maintaining the known benefits of gelatin in softgel production.
Proven in scientific studies
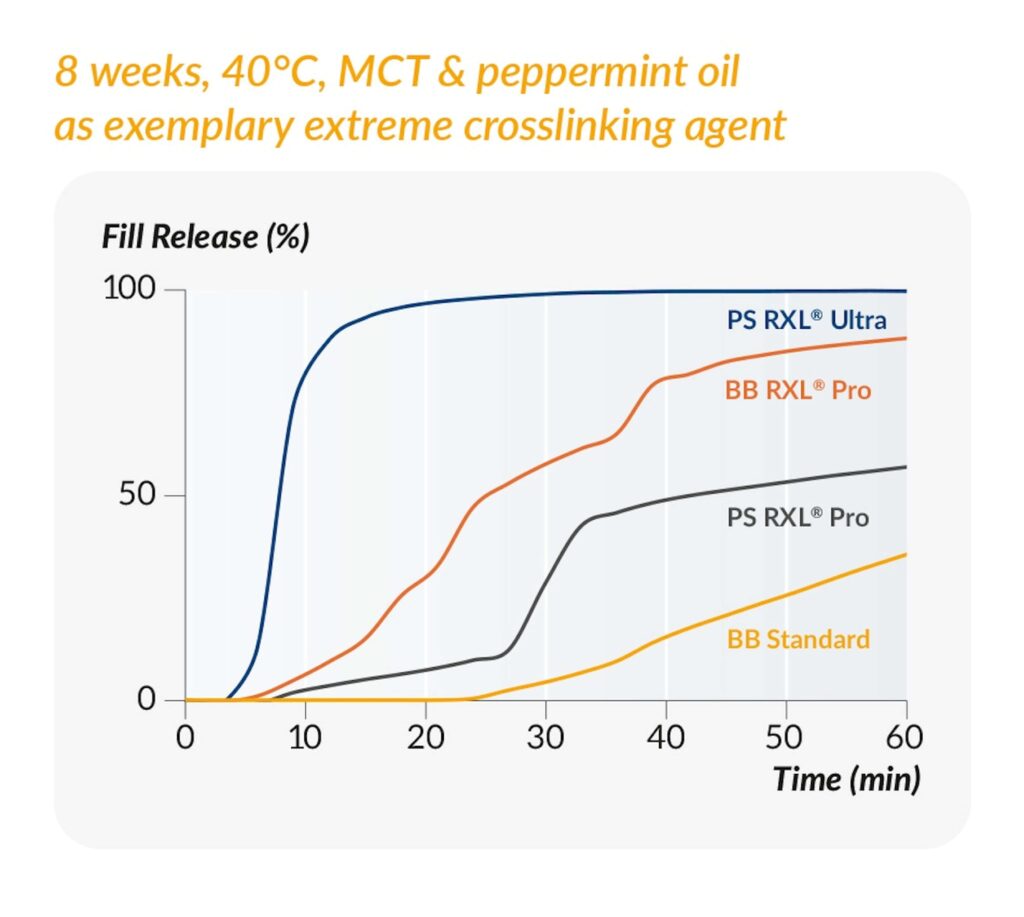
The performance of RXL® Ultra has been measured in studies comparing it with other products in the RXL® portfolio, as well as with standard gelatin. In one study, softgels were stored for eight weeks at 40°C in a medium-chain triglyceride solution containing peppermint oil, which is extremely likely to cause crosslinking.
The results show that softgels with the RXL® range outperformed the softgels made with commonly used bovine bone gelatin 160 bloom (BB Standard in the graph). The superior performance of RXL® Ultra was also demonstrated, showing no signs of crosslinking and proving excellent fill release (see Figure 1).
Another study compared the fill release of commercially available botanical softgels made from gelatin and gelatin polysuccinate – all containing potential extreme crosslinking ingredients. While the gelatin soft capsules failed to release their fill within 2 hours, gelatin polysuccinate softgels showed superior fill release performance (see Table 2, below).
Table 2: Fill release during dissolution of softgels containing ingredients with extreme crosslinking potential
| Softgel shell | Ingredient | Dissolution in HCl (simulated gastric acid) |
| Gelatin polysuccinate | Eucalyptus oil | Fill released in a few minutes |
| Lavender oil | Fill released in a few minutes | |
| Peppermint oil, Cumin oil | Fill released in a few minutes | |
| Valerian, hops | Fill released in a few minutes | |
| Gelatin | Blueberry extract | No release within 2 hours |
| Peppermint oil | No release within 2 hours | |
| Vitamin C, iron | No release within 2 hours |
Gelatin polysuccinate has a history of safe use for more than 50 years in the pharmaceutical industry in various regions, including the EU, UK, Korea and Japan. It is used in medicinal products as a softgel capsule material as well as an active pharmaceutical ingredient (API) in blood plasma expanders. However, due to the modification, it does not fit into the definition of ‘gelatin’ approved for use in food applications, so it cannot currently be used in dietary supplements. GELITA is currently working with regulators to gain approval for RXL® Ultra’s use in food supplements around the world.
In the meantime, the other three products in the RXL® portfolio are fully approved for use in food supplements in all global markets. The best RXL® choice for each application will depend on a variety of factors, including fill composition and other shell components, such as plasticizer. “We are more than happy to support supplement developers to find the most suitable solution in all cases,” says Katrin Kaspar. “And we’re looking forward to helping to encapsulate the vast potential of the natural world of botanicals in superior softgels.”
References
[1] https://pmc.ncbi.nlm.nih.gov/articles/PMC5814329/pdf/nihms939044.pdf; last accessed: 12.12.2024.
[3] https://pmc.ncbi.nlm.nih.gov/articles/PMC8659287/pdf/molecules-26-07108.pdf; last accessed: 12.12.2024.
[2] https://pmc.ncbi.nlm.nih.gov/articles/PMC4394901/; last accessed: 12.12.2024.
[4] https://pmc.ncbi.nlm.nih.gov/articles/PMC7442370/pdf/nmz065.pdf; last accessed: 12.12.2024.
[5] Domínguez R, Pateiro M, Munekata PES, McClements DJ, Lorenzo JM. Encapsulation of Bioactive Phytochemicals in Plant-Based Matrices and Application as Additives in Meat and Meat Products. Molecules. 2021 Jun 29;26(13):3984. doi: 10.3390/molecules26133984. PMID: 34210093; PMCID: PMC8272106.
[6] https://www.innovamarketinsights.com/trends/botanical-ingredients-trends/; last accessed: 10.12.2024.
[7] https://www.factmr.com/report/41/softgels-dietary-supplements-market; last accessed: 10.12.2024.
[8] https://www.futuremarketinsights.com/reports/softgel-capsules-market; last accessed: 10.12.2024.




