Ageing, which we broadly define as the time-dependent functional decline that affects most living organisms, has attracted curiosity and excited imagination throughout the history of humankind.
Ageing research has experienced an unprecedented advance over recent years, particularly with the discovery that the rate of ageing is controlled, at least to some extent, by genetic pathways and biochemical processes conserved in evolution.
The following review1 enumerates nine tentative hallmarks that represent common denominators of ageing in different organisms, with special emphasis on mammalian ageing. These hallmarks are genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, deregulated nutrient-sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, and altered intercellular communication.
A major challenge is to dissect the interconnectedness between the candidate hallmarks and their relative contribution to ageing, with the final goal of identifying pharmaceutical targets to improve human health during ageing with minimal side-effects1.
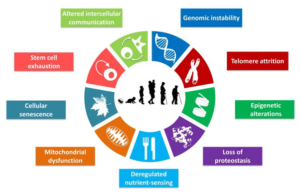
FIGURE 1. The hallmarks of aging
The scheme enumerates the nine hallmarks described in this review: genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, deregulated nutrient-sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, and altered intercellular communication.
Taken from López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013;153(6):1194-1217. doi:10.1016/j.cell.2013.05.0391
A global view of the nine candidate hallmarks of ageing enumerated in this review allows grouping them into three categories: primary, antagonistic, and integrative. (Figure 2.)
The primary hallmarks’ common characteristic is that they are all unequivocally negative. This is the case of DNA damage, including chromosomal aneuploidies, mitochondrial DNA mutations and telomere loss, epigenetic drift, and defective proteostasis.
In contrast to the primary hallmarks, antagonistic hallmarks have opposite effects depending on their intensity. At low levels, they mediate beneficial effects, but at high levels, they become deleterious. This is the case for senescence, which protects the organism from cancer, but in excess can promote ageing; similarly, reactive oxygen species (ROS) mediate cell signalling and survival, but at chronic high levels can produce cellular damage; likewise, optimal nutrient-sensing and anabolism is obviously important for survival but in excess and during time can become pathological. These hallmarks can be viewed as designed to protect the organism from damage or nutrient scarcity, but when exacerbated or chronic, they subvert their purpose and generate further damage.
A third category comprises the integrative hallmarks, stem cell exhaustion and altered intercellular communication, which directly affect tissue homeostasis and function. Notwithstanding the interconnectedness between all hallmarks, we propose some degree of hierarchical relation between them (Figure 2). The primary hallmarks could be the initiating triggers whose damaging events progressively accumulate with time. The antagonistic hallmarks, being in principle beneficial, become progressively negative in a process that is partly promoted or accelerated by the primary hallmarks.
Finally, integrative hallmarks arise when the accumulated damage caused by the primary and antagonistic hallmarks cannot be compensated by tissue homeostatic mechanisms. Because the hallmarks co-occur during ageing and are interconnected, understanding their exact causal network is an exciting challenge for future work1.
The definition of hallmarks of ageing may contribute to building a framework for future studies on the molecular mechanisms of ageing as well as for designing interventions to improve human healthspan.
FIGURE 2: Functional interconnections between the hallmarks of aging
The proposed nine hallmarks of ageing are grouped into three categories. At the top, those hallmarks are considered to be the primary causes of cellular damage. In the middle, are those considered to be part of compensatory or antagonistic responses to the damage. These responses initially mitigate the damage, but eventually, if chronic or exacerbated, they become deleterious themselves. At the bottom, there are integrative hallmarks that are the end result of the previous two groups of hallmarks and are ultimately responsible for the functional decline associated with ageing.
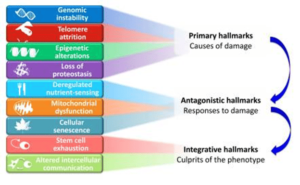
Taken from López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013;153(6):1194-1217. doi:10.1016/j.cell.2013.05.0391
Healthy ageing
Healthy ageing refers to the process of growing older in a way that maintains physical, mental, and emotional well-being and quality of life. It involves adopting a range of lifestyle practices and making informed choices to promote longevity and overall health.
Healthy ageing is not just the absence of disease but the presence of a fulfilling and purposeful life as one grows older. It involves taking a proactive approach to one’s health and well-being and making choices that support a high quality of life in the later years. It’s essential to consult with healthcare professionals for guidance and to tailor these principles to individual needs and circumstances.
Supplement’s role in healthy ageing
As we come to understand the mechanisms of ageing in the human body, it becomes easier to define potential supplements which could help and support healthy ageing. Some of them will be presented in more detail in the following text.
Blueberries
Blueberries are a popular and nutritious fruit known for their sweet taste and numerous health benefits. Blueberries are low in calories and packed with essential nutrients. They are an excellent source of vitamins and minerals, including vitamin C, vitamin K, and manganese. They are also high in dietary fiber.
Blueberries are renowned for their high levels of antioxidants, particularly anthocyanins. These antioxidants help protect the body from oxidative stress, which can lead to various health issues, including ageing and certain diseases.
Blueberries can be supplemented through a blueberry extract, isolated anthocyanins, or frozen or fresh blueberries.
The health benefits of anthocyanins have been broadly defined, particularly in the prevention of oxidative stress-related disorders, such as neurodegenerative diseases.
Bowtell et al. showed that 12 weeks of daily intake of blueberry concentrate supplementation improved the international shopping list task with no significant difference compared to the placebo among 26 healthy older adults. The primary limitation of this study was the small sample size. Thirty millilitres of blueberry concentrate were consumed once per day for 12 weeks and provided 387 mg of anthocyanidins. Chronic supplementation with blueberry concentrate providing 387 mg anthocyanins per day exerted favourable effects on cerebrovascular and cognitive function in healthy older adults. In conclusion, blueberry concentrate consumed once per day (30 mL, providing 387 mg anthocyanins) for 12 weeks increased activation of brain areas associated with cognitive processes including memory and executive function, which tend to deteriorate with age. These effects of blueberry appear to be mediated by improved vascular function as suggested by the improved resting perfusion of grey matter in the parietal and occipital lobes of the brain2.
Another study demonstrated that blueberry supplementation has neurocognitive benefits in middle-aged individuals with insulin resistance and elevated risk for future dementia. Preliminary studies indicate that blueberry supplementation can improve cognitive performance and influence metabolism and brain function and therefore may have a role in early intervention to prevent neurodegeneration.
The findings suggest that supplementation has the potential for protection against future neurocognitive decline in vulnerable individuals. Further, the level of blueberry supplementation employed in animal studies typically has not been greater than that employed in the current human trial and would be feasible for most individuals. In addition, we obtained data concerning mechanistic factors including correction of hyperinsulinemia and possibly benefits for mitochondrial function. Future research assessing blueberry supplementation over longer periods with longitudinal cognitive assessments would be valuable not only to assess its influence on the progression of cognitive decline but also to investigate further the mechanisms of neurocognitive benefit. The role of blueberry anthocyanins and proanthocyanidins in the enhancement of metabolic and mitochondrial function seems particularly salient, as these factors are intrinsic to neurodegenerative processes3.
L-carnitine
L-carnitine is an endogenous molecule and is an important contributor to cellular energy metabolism. It is present ubiquitously in the organism, and the main concentrations are found in the most active metabolic tissue, such as the myocardium and skeletal muscle. L-carnitine is indispensable for the transport of long-chain fatty acids across the inner mitochondrial membrane to their site of oxidation and the production of energy in the form of ATP. Among all the substances whose concentration decreases with age, L-carnitine diminution is fundamentally important, given its function in the production of energy. One of the most important consequences of carnitine deficiency is therefore manifested in the alteration of the metabolic pathways that lead to the production of energy.
L-carnitine is a compound produced in the body from lysine and methionine. It is also found in food, primarily in meat products, and can be taken as a supplement. L-carnitine is an amino acid derivative that plays a crucial role in the metabolism of fatty acids. Although the main dietary source of carnitine is meat, people who follow a strict plant-based diet typically do not have a higher risk of carnitine deficiency because sufficient amounts of carnitine can be synthesized in the body from other amino acids (e.g., lysine and methionine).
With age, there can be a decline in the body’s natural production of L-carnitine. This decline has led to the hypothesis that supplementing with -carnitine may help counteract some aspects of age-related metabolic changes. However, the scientific evidence supporting this claim is mixed. Mitochondrial dysfunction is a key factor in the ageing process. Some research has suggested that L-carnitine might support mitochondrial function, potentially reducing the impact of age-related cellular damage. However, the extent and significance of these effects remain uncertain.
It’s possible that L-carnitine could support brain health by aiding energy production in brain cells, but more research is needed in this area. L-carnitine has been studied in the context of cardiovascular health. Some studies suggest it may have a positive impact on factors such as blood pressure and lipid profiles, which are relevant to ageing-related cardiovascular issues. L-carnitine also possesses antioxidant properties, which could help reduce oxidative stress and cellular damage associated with ageing.
Side effects are generally rare, but some cases of upset stomachs have been reported. Carnitine and its derivatives might interact negatively with some pharmaceuticals, including anticoagulants (such as warfarin). People on thyroid medication or with hypothyroidism may also wish to forgo carnitine, as it might depress thyroid hormone levels.
The placebo-controlled, randomized, double-blind, 2-phase study that recruited. sixty-six centenarians with onset of fatigue after even slight physical activity were recruited to the study. Study results indicate that oral administration of levocarnitine produces a reduction of total fat mass, increases total muscular mass, and facilitates an increased capacity for physical and cognitive activity by reducing fatigue and improving cognitive functions4.
Protein
In seniors, higher protein intakes are associated with greater muscle mass and greater muscle mass with greater longevity and bone mineral density.
Age-related muscle loss, also known as sarcopenia, affects about 50% of males and 70% of females past the age of sixty, but it can start as early as in the twenties.
Reduced relative skeletal muscle mass in older Americans is a common occurrence that is significantly and independently associated with functional impairment and disability, particularly in older women. These observations provide strong support for the prevailing view that sarcopenia may be an important and potentially reversible cause of morbidity and mortality in older persons5.
Increased intake of protein is recommended for the elderly. In case it is not possible to get enough from food sources, it can be supplemented in powder form. Protein supplements such as whey protein, casein protein, soy protein, pea protein or protein shakes may be considered. These can help older adults meet their protein needs. Combining protein intake with resistance exercise is essential for building and maintaining muscle mass in elderly individuals with sarcopenia. Regular strength training can enhance the effects of protein intake. Sarcopenia management should be individualized, taking into account a person’s overall health, nutritional status, and specific dietary preferences and restrictions. It’s often best to consult with a healthcare professional or a registered dietitian for personalized guidance and protein dosage.

Coenzyme Q10 (CoQ10)
Coenzyme Q10 (CoQ10) is a naturally occurring compound found in every cell of the body. It plays a critical role in producing energy, as it is a key component of the electron transport chain, which is essential for generating adenosine triphosphate (ATP), the body’s primary source of cellular energy. As individuals age, the levels of CoQ10 in the body can decline, and this decline has led to an interest in the potential benefits of CoQ10 supplementation, especially in elderly individuals. CoQ10 is involved in the production of ATP, which is necessary for various cellular processes. In elderly individuals, maintaining optimal CoQ10 levels may help support energy production and combat age-related fatigue and reduced physical performance. CoQ10 has been studied for its potential role in supporting cardiovascular health. It may help improve the function of the heart and blood vessels, making it beneficial for elderly individuals who may be at greater risk of heart-related issues. Statin medications, which are commonly prescribed to lower cholesterol, can deplete CoQ10 levels in the body. Older adults who are taking statins may benefit from CoQ10 supplementation to help mitigate potential side effects.
Dose-dependence is not commonly observed with CoQ10 supplementation and 90 mg tends to be the best cost-effective dose. Higher dosages can be indicated for some conditions like cardiovascular issues.

There is growing evidence that oxidative stress is a major component of cellular senescence6. Oxidative damage in endothelial cells is proposed to play an important role in endothelial dysfunction and atherogenesis. CoQ10 could be a promising agent for reducing oxidative stress on the cellular level.
CoQ10 supplementation has been shown to have epigenetic effects in genes involved with signalling, intermediary metabolism, transport, transcription control, disease mutation, phosphorylation, and embryonal development indicating a role in the modulation of gene expression. In addition to its major function in the ETC, CoQ10 has an important anti-oxidant role in stabilizing the plasma membrane and other intracellular membranes protecting membrane phospholipids from peroxidation. Other functions include modulation of the permeability transition pore, thus playing a role in apoptosis7. CoQ10′s main functions are summarized in Figure 3.
FIGURE 3. The multiple roles of ubiquinone in the cell
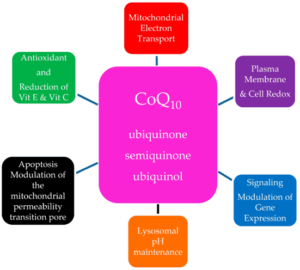
Taken from Barcelos IPd, Haas RH. CoQ10 and Aging. Biology. 2019; 8(2):28. https://doi.org/10.3390/biology80200287
There is accumulating evidence that some diseases of ageing may benefit from supplemental ubiquinol or CoQ10 treatment. Studies to date have supported the safety and the potential of CoQ10 in reducing oxidative stress biomarkers. There remains a lack of adequate large-scale clinical trials preferably utilizing ubiquinol as the better-absorbed form of CoQ10. Despite the lack of evidence, large numbers of people in the population are taking CoQ10 and other vitamins and cofactors in the hope that these agents will slow senescence and expand longevity7.
NAD/NADH (Nicotinamide Adenine Dinucleotide)
NAD (Nicotinamide Adenine Dinucleotide) is a molecule that plays a crucial role in various cellular processes, including energy production and DNA repair. It has gained significant attention in recent years for its potential role in promoting healthy ageing. Low NAD levels have been associated with a range of age-related diseases, including neurodegenerative diseases like Alzheimer’s and Parkinson’s, metabolic disorders, cardiovascular diseases, and more. Research into NAD supplementation aims to explore whether it can mitigate the risk or progression of these diseases.
The cofactor nicotinamide adenine dinucleotide (NAD) is an important metabolic regulator of cellular redox reactions and a co-factor or a co-substrate for key enzymes essential for normal cellular function in different tissues. Known as NAD+ in its oxidized state and NADH in its reduced state, it was first described more than a century ago as a molecule in the electron transport chain in the metabolic reduction-oxidation reactions in mitochondria.
Poly(ADP-ribose) polymerases (PARPs), a group of enzymes that catalyze the transfer of ADP-ribose to target proteins, use NAD as a cofactor. PARPs regulate many important cellular functions, including expression of transcription factors, gene expression and DNA repair. More recent interest in NAD emerged from research into the role of sirtuins, NAD-dependent deacylases, after the discovery that Sirtuin 2 is an NAD+-dependent histone deacetylase. Sirtuins influence many important cellular processes, including inflammation, bioenergetics, circadian rhythm generation, and cell growth, all fundamental to cellular ageing. These pathways place NAD at the center of cellular metabolism, mitochondrial function, and biological processes of ageing. Most human cells must rely on de novo creation of NAD from a variety of building blocks. NAD levels decline with increasing age.
Most researchers consider that for NAD or NAD precursors to exert beneficial effects, they must be taken up by cells to participate in pathways relevant to aging and age-related conditions. It remains to be robustly determined as to how oral supplementation of NAD produces benefits, as comparatively little work has taken place to characterize NAD+ transport into cells8.
Curcumin
Curcumin is the primary bioactive substance in turmeric. It has anti-inflammatory properties, and there is decent evidence that it can alleviate various conditions, from chronic pain to depression. Curcumin has poor bioavailability on its own, and thus it is often combined with Black Pepper or with lipids.
Curcuma longa is a flowering plant of the ginger family best known as a spice used in curry. It is a polyphenol with anti-inflammatory properties and the ability to increase the amount of antioxidants that the body produces9.

Curcumin, a polyphenol, has been shown to target multiple signalling molecules while also demonstrating activity at the cellular level, which has helped to support its multiple health benefits10. Curcumin is one of the most studied supplement ingredients in various human trials.
A controlled clinical study evaluating the health-promoting efficacy of lipidated curcumin was done in healthy middle-aged participants (40–60 years old). In this study, the participants were given either lipidated curcumin (80 mg/day) or placebo for 4 weeks. Curcumin, but not placebo, produced a decrease in salivary amylase and in the plasma levels of triglycerides, beta-amyloid, and alanine aminotransferase. Furthermore, curcumin administration in these participants increased salivary radical scavenging capacities and activities in plasma catalase, myeloperoxidase, and nitric oxide production. Overall, these results demonstrated the health-promoting effects of lipidated curcumin in healthy middle-aged people.
Curcumin, but not placebo, produced the following statistically significant changes: lowering of plasma triglyceride values, lowering of salivary amylase levels, raising of salivary radical scavenging capacities, the raising of plasma catalase activities, lowering of plasma beta-amyloid protein concentrations, lowering of plasma sICAM readings, increased plasma myeloperoxidase without increased c-reactive protein levels, increased plasma nitric oxide, and decreased plasma alanine aminotransferase activities. The conclusion is these results demonstrate that a low dose of a curcumin-lipid preparation can produce a variety of potentially health-promoting effects in healthy middle-aged people11.
An overview of the impact of curcumin on ageing and age-related diseases (ARD) at the organismal and cellular level is summarized in Figure 4.
FIGURE 4: Overview of the impact of curcumin on ageing and age-related diseases (ARD) at the organismal and cellular level.
On the organismal level, curcumin mimics caloric restriction (CR) and improves the effectiveness of physical activity (which in fact mimics CR). The potential senolytic activity of curcumin is still unclear, but curcumin can reduce inflammation and SASP, which are also considered as elements of senotherapy. Moreover, curcumin maintains the diversity of the microbiome and, in turn, the microbiota secrete β-glucuronidase, an enzyme, which, by deglucuronisation increases the level of curcumin in tissues. Curcumin is able to protect against cancer and to reduce the progression of already existing tumors. On the cellular level, curcumin elevates the level/activity of some anti-ageing proteins (e.g., sirtuins, AMPK) and inhibits pro-ageing ones (e.g., NF-κB, mTOR). Autophagy, considered as an anti-ageing mechanism, is modulated by curcumin, with the effect of preventing cell senescence. Altogether, by delaying ageing and ARD, curcumin can elongate the healthspan and probably also the lifespan.
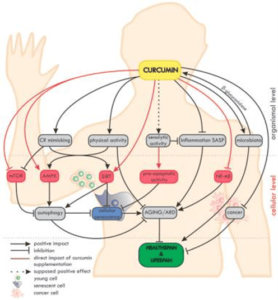
Taken from: Bielak-Zmijewska A, Grabowska W, Ciolko A, et al. The Role of Curcumin in the Modulation of Ageing. Int J Mol Sci. 2019;20(5):1239. Published 2019 Mar 12. doi:10.3390/ijms20051239
What is the future of curcumin in anti-ageing strategy? The positive impact of curcumin on ageing cannot be neglected. Undoubtedly, some precautions in curcumin exploitation necessarily take into account the biphasic response due to its hormetic properties. Curcumin applied in the diet is beneficial. It can lead to the reduction of low-grade inflammation, which is associated with ageing, and to the alleviation of symptoms of age-related diseases, including frailty. Moreover, the impact of curcumin on the microbiome seems to be very promising in the context of the modulation of the ageing process11.
Longevity
In summary, longevity refers to a long and healthy life, while Blue Zones are specific regions where people tend to live exceptionally long lives due to lifestyle and environmental factors.
Blue Zones are regions around the world where people tend to live longer and healthier lives, often well into their 90s or even 100s. These areas were identified by Dan Buettner, a National Geographic fellow, and author12. The term “Blue Zones” comes from the blue circles that Buettner used to mark these regions on a map.
Blue Zones provide valuable insights into the common habits and practices that seem to promote longevity in these areas. These include plant-based diets, regular physical activity, strong social ties, and a sense of purpose. The concept has inspired many communities and individuals to adopt these principles to improve their own health and increase their chances of living longer, healthier lives.
Factors that can contribute to longevity include:
- Genetics: Your family history and genetic makeup can play a role in how long you may live. Some individuals are genetically predisposed to live longer than others.
- Lifestyle: Maintaining a healthy lifestyle can significantly impact longevity. This includes eating a balanced diet, regular exercise, not smoking, and moderate alcohol consumption.
- Access to Healthcare: Adequate access to healthcare and regular check-ups can help catch and address health issues before they become serious.
- Mental Health: A positive mental outlook and social connections can also contribute to a longer and healthier life.
Conclusion
Food supplements can play a role in promoting longevity by providing essential nutrients and compounds that support overall health. While they are not a magic solution for living longer, they can contribute to a longer and healthier life when used in conjunction with a balanced diet and a healthy lifestyle.
References:
- 1. López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013;153(6):1194-1217. doi:10.1016/j.cell.2013.05.039
2. Joanna L. Bowtell, Zainie Aboo-Bakkar, Myra E. Conway, Anna-Lynne R. Adlam, and Jonathan Fulford. 2017. Enhanced task-related brain activation and resting perfusion in healthy older adults after chronic blueberry supplementation. Applied Physiology, Nutrition, and Metabolism. 42(7): 773-779. https://doi.org/10.1139/apnm-2016-0550
3. Krikorian R, Skelton MR, Summer SS, Shidler MD, Sullivan PG. Blueberry Supplementation in Midlife for Dementia Risk Reduction. Nutrients. 2022;14(8):1619. Published 2022 Apr 13. doi:10.3390/nu14081619
4. Malaguarnera M, Cammalleri L, Gargante MP, Vacante M, Colonna V, Motta M. L-Carnitine treatment reduces severity of physical and mental fatigue and increases cognitive functions in centenarians: a randomized and controlled clinical trial. Am J Clin Nutr. 2007;86(6):1738-1744. doi:10.1093/ajcn/86.5.1738
5. Janssen I, Heymsfield SB, Ross R. Low relative skeletal muscle mass (sarcopenia) in older persons is associated with functional impairment and physical disability. J Am Geriatr Soc. 2002;50(5):889-896. doi:10.1046/j.1532-5415.2002.50216.x
6. Huo J, Xu Z, Hosoe K, et al. Coenzyme Q10 Prevents Senescence and Dysfunction Caused by Oxidative Stress in Vascular Endothelial Cells. Oxid Med Cell Longev. 2018;2018:3181759. Published 2018 Jul 8. doi:10.1155/2018/3181759
7. Barcelos IPd, Haas RH. CoQ10 and Aging. Biology. 2019; 8(2):28. https://doi.org/10.3390/biology8020028
8. Radenkovic D, Reason, Verdin E. Clinical Evidence for Targeting NAD Therapeutically. Pharmaceuticals (Basel). 2020;13(9):247. Published 2020 Sep 15. doi:10.3390/ph13090247
9. Hewlings SJ, Kalman DS. Curcumin: A Review of Its Effects on Human Health. Foods. 2017;6(10):92. Published 2017 Oct 22. doi:10.3390/foods6100092
10. Gupta SC, Patchva S, Aggarwal BB. Therapeutic roles of curcumin: lessons learned from clinical trials. AAPS J. 2013;15(1):195-218. doi:10.1208/s12248-012-9432-8
11. Nutr J. 2012;11:79. Published 2012 Sep 26. doi:10.1186/1475-2891-11-79
12. Buettner D, Skemp S. Blue Zones: Lessons From the World’s Longest Lived. Am J Lifestyle Med. 2016;10(5):318-321. Published 2016 Jul 7. doi:10.1177/1559827616637066




